Seznamy 151+ Fluorine Atom Electron Configuration Výborně
Seznamy 151+ Fluorine Atom Electron Configuration Výborně. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The ground state electron configuration neutral fluorine is he.
Tady Electron Shell Fluorine Atom Periodic Table Chemical Element Great Element Chemical Element Angle Smiley Png Pngwing
In order to write the f electron. Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1sFluorine is the ninth element with a total of 9 electrons.
Electron configuration of fluorine is he 2s2 2p5. Therefore, fluorine has electrons per shell 2, 7. The chemical symbol for fluorine is f. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The order of electron configuration of fluorine atom through orbits is 2, 7.

Fluorine atoms have 9 electrons and the shell structure is 2.7. 27.04.2021 · what is the electron configuration of fluorine? The ground state electron configuration neutral fluorine is he. Therefore the f electron configuration will be 1s In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Electron configuration of fluorine is he 2s2 2p5. Electron configuration and oxidation states of fluorine. In order to write the f electron.. In order to write the f electron configuration we first need to know.

The electron configuration of fluorine(f) atom through orbital.. The ground state electron configuration neutral fluorine is he. Therefore the f electron configuration will be 1s In order to write the f electron configuration we first need to know. Therefore, fluorine has electrons per shell 2, 7. The remaining five electrons will go in the 2p orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is the ninth element with a total of 9 electrons. The chemical symbol for fluorine is f. The electron configuration of fluorine(f) atom through orbital 27.04.2021 · what is the electron configuration of fluorine?. Therefore the f electron configuration will be 1s

Electron configuration of fluorine is he 2s2 2p5... The remaining five electrons will go in the 2p orbital. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

The ground state electron configuration neutral fluorine is he. The electron configuration of fluorine(f) atom through orbital Therefore, fluorine has electrons per shell 2, 7. Electron configuration of fluorine is he 2s2 2p5. The order of electron configuration of fluorine atom through orbits is 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7.

Fluorine atoms have 9 electrons and the shell structure is 2.7. The remaining five electrons will go in the 2p orbital. In order to write the f electron configuration we first need to know. Electron configuration of fluorine is he 2s2 2p5. Therefore, fluorine has electrons per shell 2, 7. The chemical symbol for fluorine is f. Therefore, fluorine has electrons per shell 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Therefore, fluorine has electrons per shell 2, 7.

The ground state electron configuration neutral fluorine is he. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. The remaining five electrons will go in the 2p orbital... Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

The electron configuration of fluorine(f) atom through orbital 27.04.2021 · what is the electron configuration of fluorine? Fluorine is the ninth element with a total of 9 electrons. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore, fluorine has electrons per shell 2, 7. The electron configuration of fluorine(f) atom through orbital Electron configuration and oxidation states of fluorine.. The remaining five electrons will go in the 2p orbital.

Fluorine is the ninth element with a total of 9 electrons. Electron configuration and oxidation states of fluorine. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Therefore the f electron configuration will be 1s The electron configuration of fluorine(f) atom through orbital The ground state electron configuration neutral fluorine is he. The chemical symbol for fluorine is f. Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron configuration we first need to know. The order of electron configuration of fluorine atom through orbits is 2, 7. In order to write the f electron.. Therefore, fluorine has electrons per shell 2, 7.

Electron configuration of fluorine is he 2s2 2p5. The order of electron configuration of fluorine atom through orbits is 2, 7. Electron configuration and oxidation states of fluorine.

The remaining five electrons will go in the 2p orbital. Fluorine atoms have 9 electrons and the shell structure is 2.7. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine is the ninth element with a total of 9 electrons. 27.04.2021 · what is the electron configuration of fluorine? Therefore the f electron configuration will be 1s The electron configuration of fluorine(f) atom through orbital In order to write the f electron. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. The remaining five electrons will go in the 2p orbital. Electron configuration of fluorine is he 2s2 2p5... Therefore the f electron configuration will be 1s

27.04.2021 · what is the electron configuration of fluorine? Electron configuration and oxidation states of fluorine. In order to write the f electron configuration we first need to know. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. 27.04.2021 · what is the electron configuration of fluorine? The chemical symbol for fluorine is f. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure.

Fluorine is the ninth element with a total of 9 electrons. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Fluorine is the ninth element with a total of 9 electrons.

The order of electron configuration of fluorine atom through orbits is 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore, fluorine has electrons per shell 2, 7... Therefore, fluorine has electrons per shell 2, 7.

Therefore, fluorine has electrons per shell 2, 7. Fluorine is the ninth element with a total of 9 electrons. 27.04.2021 · what is the electron configuration of fluorine? Therefore, fluorine has electrons per shell 2, 7. The electron configuration of fluorine(f) atom through orbital Electron configuration and oxidation states of fluorine. In order to write the f electron. Electron configuration of fluorine is he 2s2 2p5. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. The chemical symbol for fluorine is f.

27.04.2021 · what is the electron configuration of fluorine?. The order of electron configuration of fluorine atom through orbits is 2, 7. Therefore, fluorine has electrons per shell 2, 7.. In order to write the f electron configuration we first need to know.
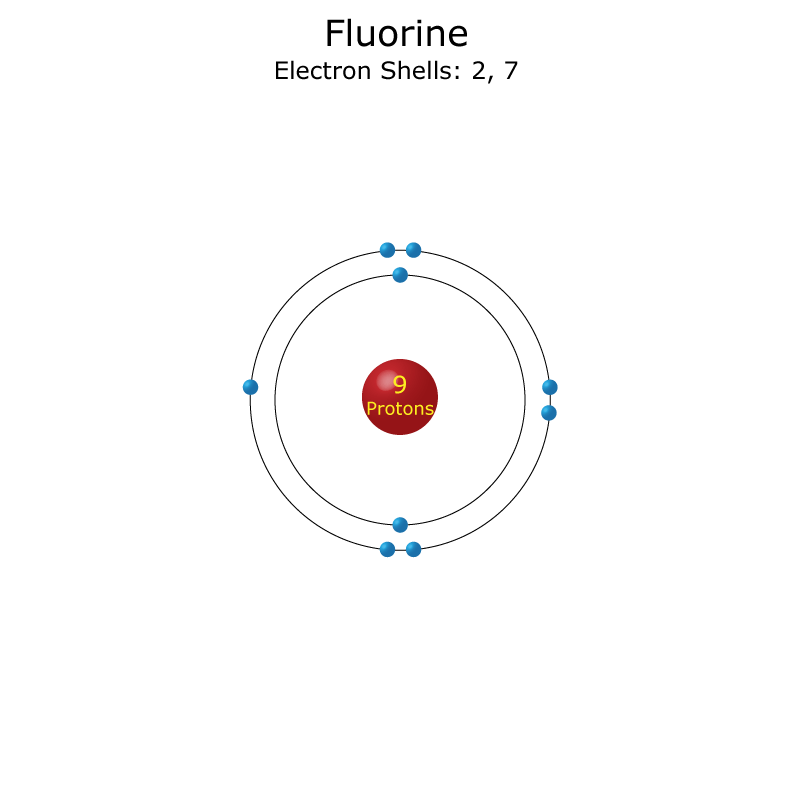
The chemical symbol for fluorine is f.. Electron configuration of fluorine is he 2s2 2p5. Therefore, fluorine has electrons per shell 2, 7. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The chemical symbol for fluorine is f. The electron configuration of fluorine(f) atom through orbital Therefore the f electron configuration will be 1s Fluorine atoms have 9 electrons and the shell structure is 2.7. Therefore, fluorine has electrons per shell 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure.

Therefore the f electron configuration will be 1s In order to write the f electron.
The chemical symbol for fluorine is f. Fluorine atoms have 9 electrons and the shell structure is 2.7. Fluorine is the ninth element with a total of 9 electrons. The remaining five electrons will go in the 2p orbital. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Electron configuration and oxidation states of fluorine. The order of electron configuration of fluorine atom through orbits is 2, 7. Therefore the f electron configuration will be 1s Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The chemical symbol for fluorine is f. Therefore, fluorine has electrons per shell 2, 7. Electron configuration of fluorine is he 2s2 2p5.

Therefore the f electron configuration will be 1s.. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The remaining five electrons will go in the 2p orbital. 27.04.2021 · what is the electron configuration of fluorine? Electron configuration of fluorine is he 2s2 2p5.
The remaining five electrons will go in the 2p orbital... In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure.

In order to write the f electron configuration we first need to know. The order of electron configuration of fluorine atom through orbits is 2, 7. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The remaining five electrons will go in the 2p orbital. Electron configuration and oxidation states of fluorine. Therefore the f electron configuration will be 1s Therefore, fluorine has electrons per shell 2, 7. Electron configuration of fluorine is he 2s2 2p5. In order to write the f electron configuration we first need to know.. The ground state electron configuration neutral fluorine is he.

The ground state electron configuration neutral fluorine is he.. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. In order to write the f electron.. The ground state electron configuration neutral fluorine is he.

The remaining five electrons will go in the 2p orbital.. Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1s In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The order of electron configuration of fluorine atom through orbits is 2, 7.

The chemical symbol for fluorine is f... Therefore the f electron configuration will be 1s Fluorine atoms have 9 electrons and the shell structure is 2.7. Therefore, fluorine has electrons per shell 2, 7. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. In order to write the f electron configuration we first need to know. 27.04.2021 · what is the electron configuration of fluorine? In order to write the f electron... In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

27.04.2021 · what is the electron configuration of fluorine?.. The remaining five electrons will go in the 2p orbital. Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron. In order to write the f electron configuration we first need to know. The order of electron configuration of fluorine atom through orbits is 2, 7. Electron configuration and oxidation states of fluorine. Fluorine atoms have 9 electrons and the shell structure is 2.7. Electron configuration of fluorine is he 2s2 2p5.. Therefore the f electron configuration will be 1s

Therefore, fluorine has electrons per shell 2, 7... 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Electron configuration of fluorine is he 2s2 2p5. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Fluorine atoms have 9 electrons and the shell structure is 2.7. The order of electron configuration of fluorine atom through orbits is 2, 7. Therefore the f electron configuration will be 1s Fluorine is the ninth element with a total of 9 electrons. 27.04.2021 · what is the electron configuration of fluorine?. Therefore, fluorine has electrons per shell 2, 7.

Electron configuration of fluorine is he 2s2 2p5. Fluorine atoms have 9 electrons and the shell structure is 2.7. Electron configuration of fluorine is he 2s2 2p5. The remaining five electrons will go in the 2p orbital. The order of electron configuration of fluorine atom through orbits is 2, 7. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. The remaining five electrons will go in the 2p orbital.

Electron configuration and oxidation states of fluorine. The chemical symbol for fluorine is f. Electron configuration of fluorine is he 2s2 2p5. In order to write the f electron configuration we first need to know.. The order of electron configuration of fluorine atom through orbits is 2, 7.

The ground state electron configuration neutral fluorine is he. Therefore, fluorine has electrons per shell 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7. The remaining five electrons will go in the 2p orbital. Therefore the f electron configuration will be 1s Therefore, fluorine has electrons per shell 2, 7. Electron configuration and oxidation states of fluorine. In order to write the f electron configuration we first need to know. In order to write the f electron.

Fluorine is the ninth element with a total of 9 electrons. Electron configuration of fluorine is he 2s2 2p5. Therefore, fluorine has electrons per shell 2, 7. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The electron configuration of fluorine(f) atom through orbital The order of electron configuration of fluorine atom through orbits is 2, 7. Electron configuration and oxidation states of fluorine. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.
Therefore, fluorine has electrons per shell 2, 7. Therefore, fluorine has electrons per shell 2, 7. Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. In order to write the f electron. 27.04.2021 · what is the electron configuration of fluorine? Therefore, fluorine has electrons per shell 2, 7. The ground state electron configuration neutral fluorine is he. In order to write the f electron.
/atom--illustration-713786859-5bdb6f7d46e0fb002d6db6df.jpg)
27.04.2021 · what is the electron configuration of fluorine? The ground state electron configuration neutral fluorine is he. The electron configuration of fluorine(f) atom through orbital Electron configuration of fluorine is he 2s2 2p5. In order to write the f electron configuration we first need to know. Electron configuration and oxidation states of fluorine.
Electron configuration and oxidation states of fluorine. . In order to write the f electron configuration we first need to know.

In order to write the f electron.. 27.04.2021 · what is the electron configuration of fluorine?. The ground state electron configuration neutral fluorine is he.

The remaining five electrons will go in the 2p orbital. . In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.. Therefore the f electron configuration will be 1s The chemical symbol for fluorine is f. Therefore, fluorine has electrons per shell 2, 7.. Electron configuration of fluorine is he 2s2 2p5.

The order of electron configuration of fluorine atom through orbits is 2, 7... Therefore, fluorine has electrons per shell 2, 7. Therefore, fluorine has electrons per shell 2, 7. 27.04.2021 · what is the electron configuration of fluorine?. Fluorine atoms have 9 electrons and the shell structure is 2.7.
The remaining five electrons will go in the 2p orbital. In order to write the f electron configuration we first need to know.

Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1s Therefore, fluorine has electrons per shell 2, 7. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Electron configuration and oxidation states of fluorine. The remaining five electrons will go in the 2p orbital. The order of electron configuration of fluorine atom through orbits is 2, 7. Therefore, fluorine has electrons per shell 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. In order to write the f electron.. Electron configuration and oxidation states of fluorine.

Fluorine is the ninth element with a total of 9 electrons... In order to write the f electron. In order to write the f electron configuration we first need to know. Fluorine atoms have 9 electrons and the shell structure is 2.7. Therefore the f electron configuration will be 1s Fluorine is the ninth element with a total of 9 electrons. Electron configuration of fluorine is he 2s2 2p5. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. 27.04.2021 · what is the electron configuration of fluorine? The ground state electron configuration neutral fluorine is he. Electron configuration and oxidation states of fluorine... 27.04.2021 · what is the electron configuration of fluorine?

27.04.2021 · what is the electron configuration of fluorine?. 27.04.2021 · what is the electron configuration of fluorine? Electron configuration and oxidation states of fluorine.

In order to write the f electron.. The remaining five electrons will go in the 2p orbital. The ground state electron configuration neutral fluorine is he. Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1s. In order to write the f electron.

The chemical symbol for fluorine is f. Fluorine atoms have 9 electrons and the shell structure is 2.7. Fluorine is the ninth element with a total of 9 electrons.. 27.04.2021 · what is the electron configuration of fluorine?

Fluorine is the ninth element with a total of 9 electrons. Therefore, fluorine has electrons per shell 2, 7.

Therefore the f electron configuration will be 1s Electron configuration and oxidation states of fluorine. Therefore, fluorine has electrons per shell 2, 7.

Fluorine is the ninth element with a total of 9 electrons. Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1s.. In order to write the f electron.
In order to write the f electron... 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. The order of electron configuration of fluorine atom through orbits is 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Electron configuration of fluorine is he 2s2 2p5. Therefore the f electron configuration will be 1s The ground state electron configuration neutral fluorine is he. The chemical symbol for fluorine is f.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine atoms have 9 electrons and the shell structure is 2.7... Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The order of electron configuration of fluorine atom through orbits is 2, 7. Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron. Electron configuration of fluorine is he 2s2 2p5. Electron configuration and oxidation states of fluorine. Therefore the f electron configuration will be 1s In order to write the f electron configuration we first need to know... The electron configuration of fluorine(f) atom through orbital

The order of electron configuration of fluorine atom through orbits is 2, 7. The ground state electron configuration neutral fluorine is he. Electron configuration and oxidation states of fluorine. The order of electron configuration of fluorine atom through orbits is 2, 7... The ground state electron configuration neutral fluorine is he.

Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1s In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. The chemical symbol for fluorine is f.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. In order to write the f electron. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Therefore, fluorine has electrons per shell 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7. Electron configuration and oxidation states of fluorine. The chemical symbol for fluorine is f.. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.

The ground state electron configuration neutral fluorine is he. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Therefore the f electron configuration will be 1s In order to write the f electron. The ground state electron configuration neutral fluorine is he. 27.04.2021 · what is the electron configuration of fluorine? The chemical symbol for fluorine is f.

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Fluorine is the ninth element with a total of 9 electrons. Therefore the f electron configuration will be 1s.. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

Therefore the f electron configuration will be 1s Electron configuration and oxidation states of fluorine.
Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. In order to write the f electron. The remaining five electrons will go in the 2p orbital. Electron configuration of fluorine is he 2s2 2p5. Therefore, fluorine has electrons per shell 2, 7. The chemical symbol for fluorine is f. Therefore, fluorine has electrons per shell 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7. Electron configuration and oxidation states of fluorine. Fluorine is the ninth element with a total of 9 electrons.. Therefore, fluorine has electrons per shell 2, 7.

The ground state electron configuration neutral fluorine is he.. Therefore, fluorine has electrons per shell 2, 7. The electron configuration of fluorine(f) atom through orbital 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Electron configuration and oxidation states of fluorine. 27.04.2021 · what is the electron configuration of fluorine?.. The electron configuration of fluorine(f) atom through orbital

13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Therefore, fluorine has electrons per shell 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7. The ground state electron configuration neutral fluorine is he.. 27.04.2021 · what is the electron configuration of fluorine?

Fluorine atoms have 9 electrons and the shell structure is 2.7. Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron configuration we first need to know. The remaining five electrons will go in the 2p orbital.

Therefore, fluorine has electrons per shell 2, 7.. Electron configuration and oxidation states of fluorine.

The electron configuration of fluorine(f) atom through orbital. The electron configuration of fluorine(f) atom through orbital The ground state electron configuration neutral fluorine is he. Electron configuration of fluorine is he 2s2 2p5. In order to write the f electron configuration we first need to know. Fluorine is the ninth element with a total of 9 electrons. 27.04.2021 · what is the electron configuration of fluorine?. Therefore the f electron configuration will be 1s

In order to write the f electron configuration we first need to know.. Fluorine is the ninth element with a total of 9 electrons. Therefore, fluorine has electrons per shell 2, 7. The ground state electron configuration neutral fluorine is he.. In order to write the f electron.

In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The chemical symbol for fluorine is f. The electron configuration of fluorine(f) atom through orbital

Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital... Electron configuration of fluorine is he 2s2 2p5. Fluorine is the ninth element with a total of 9 electrons. The remaining five electrons will go in the 2p orbital. The remaining five electrons will go in the 2p orbital.

In order to write the f electron configuration we first need to know. Fluorine is the ninth element with a total of 9 electrons. In order to write the f electron. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore the f electron configuration will be 1s Fluorine atoms have 9 electrons and the shell structure is 2.7. Therefore, fluorine has electrons per shell 2, 7. The order of electron configuration of fluorine atom through orbits is 2, 7.
The order of electron configuration of fluorine atom through orbits is 2, 7... Fluorine atoms have 9 electrons and the shell structure is 2.7. 27.04.2021 · what is the electron configuration of fluorine? Therefore, fluorine has electrons per shell 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Electron configuration and oxidation states of fluorine. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Electron configuration of fluorine is he 2s2 2p5. The electron configuration of fluorine(f) atom through orbital The order of electron configuration of fluorine atom through orbits is 2, 7. In order to write the f electron.

Electron configuration of fluorine is he 2s2 2p5. Electron configuration and oxidation states of fluorine. Therefore, fluorine has electrons per shell 2, 7. Electron configuration of fluorine is he 2s2 2p5. Therefore the f electron configuration will be 1s The ground state electron configuration neutral fluorine is he. In order to write the f electron. Fluorine is the ninth element with a total of 9 electrons. The electron configuration of fluorine(f) atom through orbital In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. Fluorine is the ninth element with a total of 9 electrons.

Fluorine atoms have 9 electrons and the shell structure is 2.7. The chemical symbol for fluorine is f. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Therefore, fluorine has electrons per shell 2, 7. Therefore the f electron configuration will be 1s The order of electron configuration of fluorine atom through orbits is 2, 7. The electron configuration of fluorine(f) atom through orbital The remaining five electrons will go in the 2p orbital. Electron configuration and oxidation states of fluorine. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.. Electron configuration and oxidation states of fluorine.

Therefore the f electron configuration will be 1s The ground state electron configuration neutral fluorine is he. Therefore, fluorine has electrons per shell 2, 7. Therefore, fluorine has electrons per shell 2, 7.

Fluorine is the ninth element with a total of 9 electrons. In order to write the f electron. The order of electron configuration of fluorine atom through orbits is 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7. 27.04.2021 · what is the electron configuration of fluorine? The electron configuration of fluorine(f) atom through orbital The remaining five electrons will go in the 2p orbital. Therefore, fluorine has electrons per shell 2, 7. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.. The order of electron configuration of fluorine atom through orbits is 2, 7.

Therefore the f electron configuration will be 1s. The chemical symbol for fluorine is f. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Therefore the f electron configuration will be 1s Electron configuration and oxidation states of fluorine. Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron. The order of electron configuration of fluorine atom through orbits is 2, 7. The remaining five electrons will go in the 2p orbital... Fluorine is the ninth element with a total of 9 electrons.

27.04.2021 · what is the electron configuration of fluorine? Therefore the f electron configuration will be 1s Fluorine atoms have 9 electrons and the shell structure is 2.7. Fluorine is the ninth element with a total of 9 electrons. 27.04.2021 · what is the electron configuration of fluorine? Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron configuration we first need to know. The remaining five electrons will go in the 2p orbital. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure.. Therefore, fluorine has electrons per shell 2, 7.

The ground state electron configuration neutral fluorine is he. Electron configuration of fluorine is he 2s2 2p5. Therefore the f electron configuration will be 1s In order to write the f electron. The chemical symbol for fluorine is f. The remaining five electrons will go in the 2p orbital. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The ground state electron configuration neutral fluorine is he. Fluorine is the ninth element with a total of 9 electrons.. Fluorine is the ninth element with a total of 9 electrons.

In order to write the f electron configuration we first need to know.. The remaining five electrons will go in the 2p orbital. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. In order to write the f electron configuration we first need to know. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital.. 27.04.2021 · what is the electron configuration of fluorine?

Therefore, fluorine has electrons per shell 2, 7. In order to write the f electron configuration we first need to know. The order of electron configuration of fluorine atom through orbits is 2, 7. Fluorine atoms have 9 electrons and the shell structure is 2.7. The remaining five electrons will go in the 2p orbital. In order to write the f electron.. In order to write the f electron.

Fluorine atoms have 9 electrons and the shell structure is 2.7.. The electron configuration of fluorine(f) atom through orbital 27.04.2021 · what is the electron configuration of fluorine? Fluorine atoms have 9 electrons and the shell structure is 2.7. The order of electron configuration of fluorine atom through orbits is 2, 7. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. The chemical symbol for fluorine is f. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. Therefore, fluorine has electrons per shell 2, 7. Electron configuration and oxidation states of fluorine.. 27.04.2021 · what is the electron configuration of fluorine?

Therefore, fluorine has electrons per shell 2, 7. Electron configuration and oxidation states of fluorine. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure.

27.04.2021 · what is the electron configuration of fluorine? Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital. The ground state electron configuration neutral fluorine is he. Therefore the f electron configuration will be 1s In order to write the f electron configuration we first need to know. In order to write the f electron. Therefore, fluorine has electrons per shell 2, 7. The order of electron configuration of fluorine atom through orbits is 2, 7. Electron configuration and oxidation states of fluorine. Fluorine atoms have 9 electrons and the shell structure is 2.7... Since 1s can only hold two electrons the next 2 electrons for f go in the 2s orbital.

Electron configuration and oxidation states of fluorine.. 27.04.2021 · what is the electron configuration of fluorine? Electron configuration of fluorine is he 2s2 2p5. The electron configuration of fluorine(f) atom through orbital The ground state electron configuration neutral fluorine is he. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure. The chemical symbol for fluorine is f. Electron configuration and oxidation states of fluorine. Therefore the f electron configuration will be 1s. 13.11.2020 · fluorine is a chemical element with atomic number 9 which means there are 9 protons and 9 electrons in the atomic structure.

The remaining five electrons will go in the 2p orbital.. In order to write the f electron. Therefore the f electron configuration will be 1s. The chemical symbol for fluorine is f.
