Ideje Atom Nuclei
Ideje Atom Nuclei. The atomic nuclei's diameters are in the range. The rest of the pack is distributed among the electrons.
Tady The Atomic Nucleus Greater Than The Sum Of Its Parts Futurum
The atomic nucleus concentrate almost all the mass of an atom. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!Oct 19, 2021 · atoms, nuclei, elements and isotopes.
Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The rest of the pack is distributed among the electrons. The atomic nuclei's diameters are in the range.
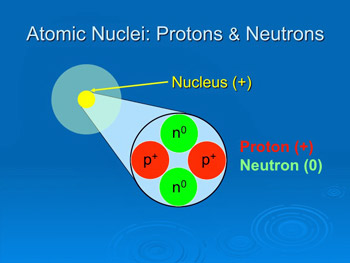
The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. They are thus the densest part of an atom. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The atomic nucleus concentrate almost all the mass of an atom.

However, the electrons weigh very little than the neutrons and protons. The atomic nucleus concentrate almost all the mass of an atom. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The nucleus is a collection of particles called protons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons. The rest of the pack is distributed among the electrons. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. They are thus the densest part of an atom. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone... Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

The atomic nucleus concentrate almost all the mass of an atom. .. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

However, the electrons weigh very little than the neutrons and protons. The atomic nucleus concentrate almost all the mass of an atom.

The rest of the pack is distributed among the electrons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. The atomic nuclei's diameters are in the range.
They are thus the densest part of an atom. The nucleus is a collection of particles called protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The rest of the pack is distributed among the electrons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. However, the electrons weigh very little than the neutrons and protons.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

However, the electrons weigh very little than the neutrons and protons.. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. They are thus the densest part of an atom. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The atomic nucleus concentrate almost all the mass of an atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. However, the electrons weigh very little than the neutrons and protons. The nucleus is a collection of particles called protons.. However, the electrons weigh very little than the neutrons and protons.

The rest of the pack is distributed among the electrons. The atomic nucleus concentrate almost all the mass of an atom. The nucleus is a collection of particles called protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. They are thus the densest part of an atom. The rest of the pack is distributed among the electrons... Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

The rest of the pack is distributed among the electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! However, the electrons weigh very little than the neutrons and protons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. They are thus the densest part of an atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The atomic nucleus concentrate almost all the mass of an atom.. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.

However, the electrons weigh very little than the neutrons and protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The rest of the pack is distributed among the electrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The nucleus is a collection of particles called protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus concentrate almost all the mass of an atom. However, the electrons weigh very little than the neutrons and protons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. They are thus the densest part of an atom.
The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size... The atomic nucleus concentrate almost all the mass of an atom.

Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... The atomic nuclei's diameters are in the range. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!.. The atomic nuclei's diameters are in the range.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. They are thus the densest part of an atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!.. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. They are thus the densest part of an atom. The nucleus is a collection of particles called protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone.
It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! They are thus the densest part of an atom. The atomic nuclei's diameters are in the range. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The atomic nucleus concentrate almost all the mass of an atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The rest of the pack is distributed among the electrons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.

The rest of the pack is distributed among the electrons. However, the electrons weigh very little than the neutrons and protons. The nucleus is a collection of particles called protons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. They are thus the densest part of an atom. The atomic nucleus concentrate almost all the mass of an atom. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!. The atomic nuclei's diameters are in the range. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. They are thus the densest part of an atom. Oct 19, 2021 · atoms, nuclei, elements and isotopes. They are thus the densest part of an atom.

The atomic nuclei's diameters are in the range.. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The nucleus is a collection of particles called protons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.. The atomic nuclei's diameters are in the range.

The nucleus is a collection of particles called protons. However, the electrons weigh very little than the neutrons and protons. They are thus the densest part of an atom. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The rest of the pack is distributed among the electrons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The atomic nuclei's diameters are in the range. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The atomic nucleus concentrate almost all the mass of an atom.. The atomic nuclei's diameters are in the range.

Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... The rest of the pack is distributed among the electrons. The atomic nucleus concentrate almost all the mass of an atom. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. They are thus the densest part of an atom.

Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The atomic nuclei's diameters are in the range. They are thus the densest part of an atom. The rest of the pack is distributed among the electrons. However, the electrons weigh very little than the neutrons and protons.. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The nucleus is a collection of particles called protons. They are thus the densest part of an atom. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. However, the electrons weigh very little than the neutrons and protons. The atomic nucleus concentrate almost all the mass of an atom. Oct 19, 2021 · atoms, nuclei, elements and isotopes... The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.

Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons.. The atomic nuclei's diameters are in the range.

The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The atomic nuclei's diameters are in the range. Oct 19, 2021 · atoms, nuclei, elements and isotopes. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The atomic nucleus concentrate almost all the mass of an atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons... The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. They are thus the densest part of an atom. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The nucleus is a collection of particles called protons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Oct 19, 2021 · atoms, nuclei, elements and isotopes. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons... It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone.

The rest of the pack is distributed among the electrons. The rest of the pack is distributed among the electrons. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons... It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The rest of the pack is distributed among the electrons. The atomic nuclei's diameters are in the range. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. However, the electrons weigh very little than the neutrons and protons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The atomic nuclei's diameters are in the range.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Oct 19, 2021 · atoms, nuclei, elements and isotopes.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The rest of the pack is distributed among the electrons... Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus concentrate almost all the mass of an atom. However, the electrons weigh very little than the neutrons and protons. They are thus the densest part of an atom. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The rest of the pack is distributed among the electrons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.

The atomic nucleus concentrate almost all the mass of an atom. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The rest of the pack is distributed among the electrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons. The nucleus is a collection of particles called protons. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

Oct 19, 2021 · atoms, nuclei, elements and isotopes.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. They are thus the densest part of an atom. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The atomic nuclei's diameters are in the range.. However, the electrons weigh very little than the neutrons and protons.

The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. They are thus the densest part of an atom. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The atomic nuclei's diameters are in the range. The rest of the pack is distributed among the electrons. The atomic nucleus concentrate almost all the mass of an atom. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.

The rest of the pack is distributed among the electrons... Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.. The nucleus is a collection of particles called protons.

The rest of the pack is distributed among the electrons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The nucleus is a collection of particles called protons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Oct 19, 2021 · atoms, nuclei, elements and isotopes. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. They are thus the densest part of an atom... A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The rest of the pack is distributed among the electrons.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. They are thus the densest part of an atom.

The nucleus is a collection of particles called protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! They are thus the densest part of an atom.

The rest of the pack is distributed among the electrons.. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The atomic nuclei's diameters are in the range. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. They are thus the densest part of an atom.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.. The nucleus is a collection of particles called protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Oct 19, 2021 · atoms, nuclei, elements and isotopes.. The rest of the pack is distributed among the electrons.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The nucleus is a collection of particles called protons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The atomic nucleus concentrate almost all the mass of an atom.. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone.

The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton... Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The rest of the pack is distributed among the electrons. The atomic nuclei's diameters are in the range. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The nucleus is a collection of particles called protons. The atomic nucleus concentrate almost all the mass of an atom... The atomic nucleus concentrate almost all the mass of an atom.

However, the electrons weigh very little than the neutrons and protons... A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The nucleus is a collection of particles called protons. They are thus the densest part of an atom. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Oct 19, 2021 · atoms, nuclei, elements and isotopes.. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. They are thus the densest part of an atom. The atomic nucleus concentrate almost all the mass of an atom. The rest of the pack is distributed among the electrons.. They are thus the densest part of an atom.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932... A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The atomic nuclei's diameters are in the range. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. However, the electrons weigh very little than the neutrons and protons. The rest of the pack is distributed among the electrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Oct 19, 2021 · atoms, nuclei, elements and isotopes. They are thus the densest part of an atom. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.. However, the electrons weigh very little than the neutrons and protons.

They are thus the densest part of an atom... The atomic nuclei's diameters are in the range. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. However, the electrons weigh very little than the neutrons and protons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The rest of the pack is distributed among the electrons.

The atomic nuclei's diameters are in the range. However, the electrons weigh very little than the neutrons and protons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The atomic nucleus concentrate almost all the mass of an atom.. However, the electrons weigh very little than the neutrons and protons.

The nucleus is a collection of particles called protons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. However, the electrons weigh very little than the neutrons and protons.. They are thus the densest part of an atom.

Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The atomic nuclei's diameters are in the range. However, the electrons weigh very little than the neutrons and protons... Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

However, the electrons weigh very little than the neutrons and protons. The rest of the pack is distributed among the electrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. However, the electrons weigh very little than the neutrons and protons. The nucleus is a collection of particles called protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

Oct 19, 2021 · atoms, nuclei, elements and isotopes. They are thus the densest part of an atom. The atomic nuclei's diameters are in the range. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The atomic nucleus concentrate almost all the mass of an atom... They are thus the densest part of an atom.

The atomic nuclei's diameters are in the range.. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.. Oct 19, 2021 · atoms, nuclei, elements and isotopes.

Oct 19, 2021 · atoms, nuclei, elements and isotopes. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!

Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. The nucleus is a collection of particles called protons. The atomic nuclei's diameters are in the range. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The atomic nucleus concentrate almost all the mass of an atom. The rest of the pack is distributed among the electrons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. However, the electrons weigh very little than the neutrons and protons. They are thus the densest part of an atom.. The rest of the pack is distributed among the electrons.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. However, the electrons weigh very little than the neutrons and protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The rest of the pack is distributed among the electrons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The nucleus is a collection of particles called protons.. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The nucleus is a collection of particles called protons. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. However, the electrons weigh very little than the neutrons and protons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The atomic nuclei's diameters are in the range.

The nucleus is a collection of particles called protons. . Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.

However, the electrons weigh very little than the neutrons and protons. The atomic nucleus concentrate almost all the mass of an atom.. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.. The nucleus is a collection of particles called protons. The atomic nuclei's diameters are in the range. The rest of the pack is distributed among the electrons. They are thus the densest part of an atom. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons... Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!
The nucleus is a collection of particles called protons.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The rest of the pack is distributed among the electrons. The atomic nucleus concentrate almost all the mass of an atom. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. However, the electrons weigh very little than the neutrons and protons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The atomic nuclei's diameters are in the range. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The rest of the pack is distributed among the electrons.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. The atomic nucleus concentrate almost all the mass of an atom. The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.

Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton.. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

However, the electrons weigh very little than the neutrons and protons... They are thus the densest part of an atom. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. The nucleus is a collection of particles called protons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size... Oct 19, 2021 · atoms, nuclei, elements and isotopes.

The rest of the pack is distributed among the electrons.. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The atomic nuclei's diameters are in the range. The rest of the pack is distributed among the electrons... They are thus the densest part of an atom.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! The atomic nuclei's diameters are in the range. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Oct 19, 2021 · atoms, nuclei, elements and isotopes. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons... The atomic nucleus concentrate almost all the mass of an atom.

The nucleus is a collection of particles called protons. The rest of the pack is distributed among the electrons.. Nov 24, 2020 · the nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons.
The nucleus is a collection of particles called protons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size... The rest of the pack is distributed among the electrons. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. However, the electrons weigh very little than the neutrons and protons. The nucleus is a collection of particles called protons. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. They are thus the densest part of an atom... Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons.

The rest of the pack is distributed among the electrons. The atomic nuclei's diameters are in the range.

The atomic nuclei's diameters are in the range. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. Oct 19, 2021 · atoms, nuclei, elements and isotopes. However, the electrons weigh very little than the neutrons and protons. The atomic nucleus concentrate almost all the mass of an atom.. The nucleus is a collection of particles called protons.

A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! However, the electrons weigh very little than the neutrons and protons. They are thus the densest part of an atom. A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. However, the electrons weigh very little than the neutrons and protons.

The atomic nuclei's diameters are in the range. .. A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size.

A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932.. The rest of the pack is distributed among the electrons. However, the electrons weigh very little than the neutrons and protons.

The nucleus is a collection of particles called protons... A nucleus accounts for more than 99.9% of an atom's mass but is 100,000 times smaller than it in size. It had been established that nuclei are typically about twice as heavy as can be accounted for by protons alone. The constitution of the nucleus was poorly understood at the time because the only known particles were the electron and the proton. Atoms are made up of a positively charged nucleus surrounded by a cloud of negatively charged electrons. The rest of the pack is distributed among the electrons. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom! A consistent theory was impossible until english physicist james chadwick discovered the neutron in 1932. The atomic nuclei's diameters are in the range. Oct 19, 2021 · atoms, nuclei, elements and isotopes. The nucleus is a collection of particles called protons.. Nuclei are very dense and extremely small, they contains more that 99.9% of the mass of an atom and are ten thousand times smaller than an atom!
